How to Verify Research Peptides: COAs, Batch Numbers, and Third-Party Testing
As the research peptide market continues to grow, one factor increasingly separates reliable suppliers from questionable ones: testing transparency and batch traceability.
Researchers rely on accurate documentation to verify the identity and purity of materials used in laboratory environments. Without a clear connection between testing reports and the material being supplied, it can be difficult to determine whether documentation truly reflects the product received.
Understanding how batch numbers, third-party laboratory testing, and Certificate of Analysis (COA) verification work can help researchers make more informed sourcing decisions.
Researchers often review documentation before selecting a supplier to ensure materials are properly verified and traceable.
https://apexpeptidesupply.com/products/ – Research Peptide Catalog]
What Is a Batch Number?
A batch number (sometimes called a lot number) is a unique identifier assigned to a specific production run of a product.
Batch numbers allow materials to be tracked through several stages of the supply chain, including:
• manufacturing
• quality testing
• packaging
• distribution
By assigning a unique identifier to each production run, suppliers create a system that links the product being sold with the documentation used to verify it.
Without batch identification, it becomes difficult to confirm whether testing documentation actually corresponds to the product being supplied.
Researchers often compare the batch number printed on the vial with the batch number listed on the testing documentation.
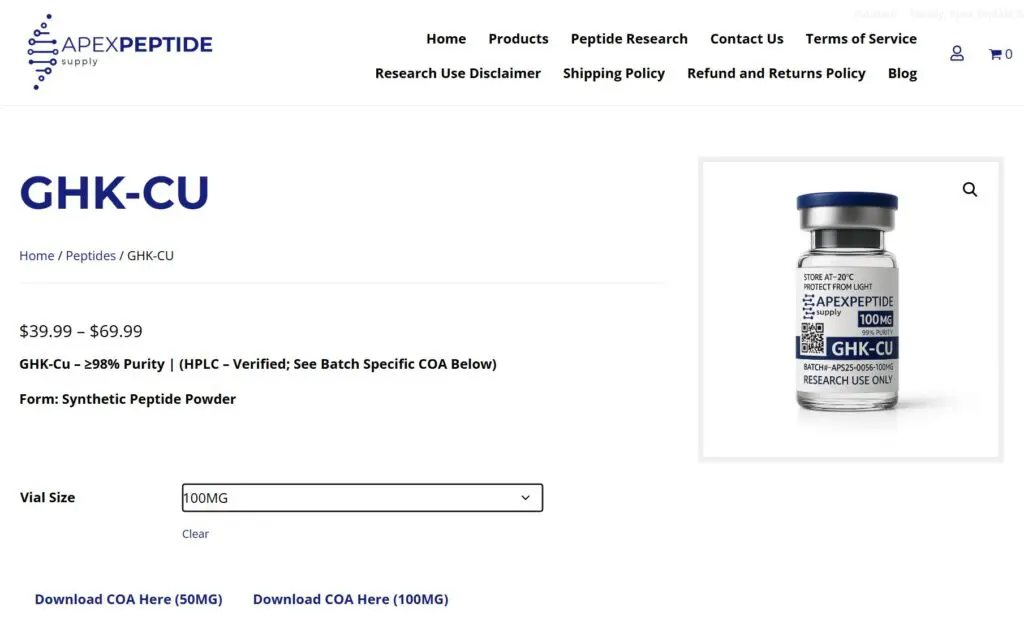
[https://apexpeptidesupply.com/product/ghk-cu/ – Example Product Page]
The Role of Third-Party Laboratory Testing
Independent laboratory testing plays an important role in verifying peptide identity and purity.
Two common analytical methods used for peptide verification include:
HPLC (High-Performance Liquid Chromatography)
HPLC is used to analyze peptide purity and detect impurities present within a sample.
LC-MS (Liquid Chromatography–Mass Spectrometry)
LC-MS confirms molecular identity by verifying that the molecular mass of the compound matches the expected peptide structure.
When testing is performed by independent laboratories and linked directly to a specific batch number, researchers can confirm that the material they receive corresponds to the documented analysis.
Transparent research peptide suppliers make these testing reports available so researchers can review the documentation associated with each batch.
Why Generic COAs Can Be Problematic
Some suppliers publish a single generic Certificate of Analysis for a product regardless of when the material was produced.
While this may appear informative, it creates uncertainty if the report is not tied to a specific batch.
In transparent supply chains, each production batch should have its own batch-specific Certificate of Analysis that corresponds directly to the material produced during that production run.
Batch-specific documentation helps ensure that the testing report accurately represents the material associated with that batch.
The Importance of Traceability
Traceability creates a documented connection between each stage of the supply process:
Manufacturing → Testing → Distribution → Final Product
This chain of documentation allows researchers to verify that the testing documentation corresponds to the exact material being used in their work.
In many scientific supply industries, traceability like this is considered a standard part of responsible documentation.
Suppliers that provide batch-specific testing documentation and clear verification systems help improve transparency within the research peptide industry.
COA Verification Through QR Codes
Some suppliers now provide QR code access to batch testing documentation, allowing researchers to quickly verify testing information.
With QR-based verification systems, researchers can scan a code associated with a batch and view the corresponding Certificate of Analysis directly.
This type of verification system can help:
• simplify access to testing documentation
• reduce the risk of outdated reports being shared
• allow faster verification of batch information
Accessible documentation makes it easier for researchers to review testing reports associated with the materials they receive.

The Apex Peptide Supply Approach to Batch Verification
At Apex Peptide Supply, transparency and traceability are built into how research materials are documented.
Each production batch is assigned a unique APS batch number that connects the product to its testing documentation.
Our verification process includes:
• assigning a unique APS batch number to each production lot
• independent third-party laboratory testing
• batch-specific Certificates of Analysis
• documentation accessible directly from product pages
Researchers can review testing documentation before purchasing or after receiving their product.
How to Access COAs on Apex Peptide Supply
Certificates of Analysis are available directly within the product pages on our website.
COAs can be downloaded here:
[https://apexpeptidesupply.com/wp-content/uploads/2026/02/APS26-0098-GHK-CU100MG-BioV.pdf]
To access the correct Certificate of Analysis for a specific product:
- Navigate to the product page for the research material you are reviewing.
- Select the specific mg size you are researching.
- Once the mg option is selected, a COA link will appear for that exact batch.
This link will take you to the Certificate of Analysis associated with the batch tied to that specific product variation.
Each COA includes:
• the batch number
• third-party testing results
• analytical methods used
• a photo of the vial tested
The batch number shown on the COA matches:
• the batch number printed on the product label
• the batch number shown in the COA document
• the batch number visible on the vial in the COA photo
This allows researchers to verify that the batch number on the product they receive corresponds directly with the testing documentation and the vial shown in the laboratory report.
When the product arrives, researchers can compare the batch number on the vial they are holding with the batch number shown on the COA and the vial photo included in the testing report.
This verification system helps maintain a clear connection between the tested material and the product received.
👉
“Learn more about BPC-157 here” → https://apexpeptidesupply.com/bpc-157-research-peptide/
What to Look for When Reviewing a COA
When verifying a Certificate of Analysis, researchers often review several key details:
• batch number matching the product
• laboratory name and testing date
• analytical methods used (HPLC, LC-MS, etc.)
• reported purity levels
Matching the batch number on the product with the batch number listed on the COA helps confirm that the testing documentation corresponds to the correct material.
Frequently Asked Questions About Peptide Testing
Why are batch numbers important for research peptides?
Batch numbers connect a product to its specific testing documentation. This allows researchers to confirm that the Certificate of Analysis corresponds to the exact material being supplied.
What information is included in a Certificate of Analysis?
A COA typically includes testing results such as purity analysis, analytical methods used, molecular identity confirmation, and the batch number associated with the tested material.
How do researchers verify that a COA matches the product they receive?
Researchers compare the batch number printed on the vial with the batch number listed on the COA documentation. Some suppliers also include a vial photo in the COA to help confirm the batch that was tested.
Why is third-party testing important?
Independent laboratory testing provides an objective analysis of the material and helps verify peptide identity and purity.
Final Thoughts
As the research peptide market continues to expand, testing transparency and batch traceability are becoming increasingly important.
Batch numbers, independent laboratory testing, and accessible Certificates of Analysis allow researchers to verify that materials are properly documented and traceable.
Reviewing this information before sourcing research materials can help improve confidence in the documentation associated with those materials.
Researchers who prioritize verification and traceability often review available testing documentation before selecting a supplier.
[ https://apexpeptidesupply.com/– Homepage Verification Section]
Link this to your homepage once you add the Verification & Transparency section.
